Dishwashing liquids have become an indispensable part of our daily routines. We rely on these essential cleaning products to tackle stubborn grease, grime, and food residue on our dishes, pots, and pans. Have you ever wondered what makes these dishwashing liquids so effective in cutting through tough stains? The answer lies in the surfactants they contain.
What Are Surfactants?
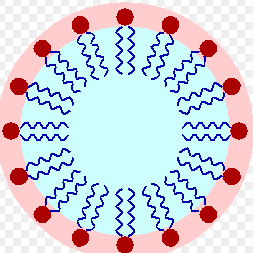
Surfactants, short for surface active agents, are key ingredients found in many household cleaners, including dishwashing liquids. These chemical compounds are responsible for reducing the surface tension of water, allowing it to spread more easily and penetrate the dirt and grease on our dishes.
Surfactants consist of molecules that have two main components: a hydrophilic (water-attracting) head and a hydrophobic (water-repelling) tail. It is this unique molecular structure that enables them to interact with both water and oils, facilitating the removal of grease and food residue in the process of dishwashing.
Types of Surfactants:
Anionic Surfactants:
Anionic surfactants are the most commonly used type in dishwashing liquids. These surfactants have negative charges on their hydrophilic heads, which allow them to attract and surround positively charged ions, such as calcium and magnesium ions, found in hard water. By forming complexes with these ions, anionic surfactants prevent them from interfering with the cleaning process.
Sodium lauryl sulfate (SLS) and sodium laureth sulfate (SLES) are two examples of anionic surfactants commonly found in dishwashing liquids. They are highly effective in breaking down grease and grime, ensuring squeaky clean dishes with each wash.
Nonionic Surfactants:
Nonionic surfactants are another type commonly used in dishwashing liquids. Unlike anionic surfactants, nonionic surfactants do not carry a charge, making them less likely to react with hard water ions. Instead, they rely on their hydrophobic tails to dissolve and remove grease and oils.
Due to their gentle nature and low toxicity, nonionic surfactants are often used in dishwashing liquids that are advertised as being safe for both our hands and the environment. They provide effective cleaning power while being less harsh on our skin and aquatic ecosystems.
Cationic and Amphoteric Surfactants:
Cationic surfactants carry positive charges on their hydrophilic heads and are rarely used in dishwashing liquids due to their high propensity for causing irritation. They are more commonly found in fabric softeners and disinfectants.
Amphoteric surfactants, on the other hand, possess both positive and negative charges and can act as either anionic or cationic surfactants, depending on the conditions. Although they are not widely used in dishwashing liquids, they can be found in some specialized products designed for sensitive skin.
The Role of Surfactants in Dishwashing Liquids:
Surfactants play a crucial role in dishwashing liquids by reducing the surface tension of water. By doing so, they enable water to spread across the surface of dirty dishes, allowing it to penetrate and dislodge the grease, food particles, and other residues.
This is achieved through a phenomenon known as emulsification. Surfactants dissolve in water and surround oily substances, breaking them down into smaller droplets called micelles. These micelles are dispersed in the water and can be easily rinsed away, leaving dishes clean and free from grease.
Enhancing the Performance of Dishwashing Liquids:
In addition to surfactants, dishwashing liquids often contain other ingredients that further enhance their cleaning performance. These may include:
Enzymes:
Enzymes, such as proteases and amylases, are added to dishwashing liquids to break down food proteins and starches, respectively. By targeting specific types of residues, enzymes enhance the overall stain-fighting capabilities of the detergent.
Fragrances:
Fragrances are incorporated into dishwashing liquids to improve the overall sensory experience during dishwashing. While not directly involved in the cleaning process, pleasant scents can contribute to a more enjoyable chore and leave a fresh aroma on your dishes.
Preservatives:
To ensure the shelf life and prevent bacterial growth, dishwashing liquids may contain preservatives. These ingredients help maintain the effectiveness and integrity of the product over time.
In Conclusion
Surfactants are the powerhouses behind the effectiveness of dishwashing. Anionic surfactants, such as sodium lauryl sulfate and sodium laureth sulfate, are commonly used to break down grease and grime, while nonionic surfactants provide a milder option suitable for sensitive skin and the environment.
Next time you tackle a stack of dirty dishes, you can appreciate the role that surfactants play in making your cleaning task a breeze. With their ability to reduce water’s surface tension and tackle stubborn stains, surfactants ensure your dishes come out sparkling clean after each wash.
